Fluorescence Filters for Microscopy and Imaging
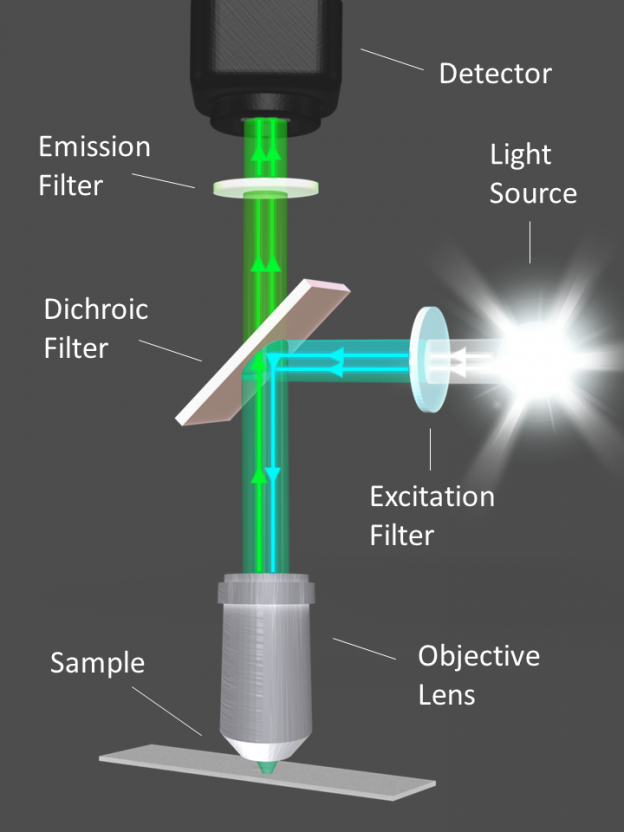
Figure 1. Diagram illustrating the optical filters and light path of a fluorescence microscope.
Image credit: Alluxa
Fluorescence microscopes and imaging systems utilize fluorescent biomarkers and fluorescence filter sets to create bright, high contrast images of biomolecules, organelles, cells, tissues, organs, and organ systems. Because image quality is highly dependent on the design and overall performance of the fluorescence filters integrated into these systems, optical filter performance is just as important to the final image as sample preparation and fluorophore selection.
Fluorescence
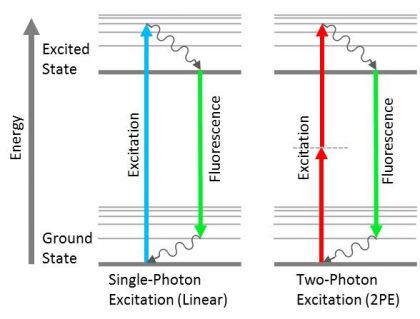
When a photon of light at a specific wavelength is absorbed by an atom or molecule, an orbital electron jumps from its ground state (S0) to an excited singlet state (S1). As the electron returns to its ground state, a photon is emitted that has lower energy, and therefore a longer wavelength, than the photon that was initially absorbed (Figure 2). This phenomenon, known as fluorescence, is the key concept behind a large variety of instruments used in biological science and medicine. Fluorescence microscopes, imaging systems, flow cytometers, and DNA sequencers all use a light source, such as a laser, LED, or broadband lamp, to excite the fluorescent tags within a sample. Fluorescence filters are used to isolate and direct the excitation signal to the sample and the emission signal to the detector or eyepiece (Figure 1).
In some cases, multiple longwave photons can be simultaneously absorbed by a single atom or molecule, resulting in an emitted photon that has higher energy, and a shorter wavelength, than those that were initially absorbed (Figure 2). This non-linear fluorescence response is employed in two-photon and multi-photon fluorescence microscopy systems. However, an electron in an excited state can also return to the ground state by other processes also utilized in microscopy; these include fluorescence resonance energy transfer (FRET) and stimulated emission depletion (STED).
Figure 2. Jablonski diagrams showing the difference between linear fluorescence (left) and two-photon excitation (right).
Fluorescent Labels
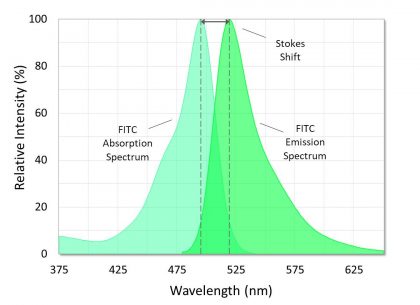
Samples studied using fluorescence are generally labeled with, or naturally contain, substances called fluorophores (or fluorochromes) that give off a relatively strong emission signal as a property of their aromatic chemical structures. All fluorophores have a unique absorption (or excitation) spectrum and a unique emission spectrum, with each peaking in energy or intensity at a specific wavelength. The wavelength difference between the peak absorption and peak emission of a given fluorophore is known as the Stokes shift (Figure 3), which can range from just a few, to several hundred nanometers.
Figure 3. Absorption and emission spectra of fluorescein isothiocyanate (FITC). The Stokes shift is the difference between the spectral peaks.
A large variety of fluorescent proteins, probes, dyes, and other fluorophores are available for use as fluorescent labels. Each of these has different excitation and emission spectra and a different Stokes shift, and all vary in terms of brightness, maturation time, and photostability. Additionally, emission intensity depends on the wavelength of light absorbed by the fluorophore; light corresponding to peak absorption will result in maximum emission intensity, while light at wavelengths farther from the absorption peak will produce a lower-intensity emission signal. Emission signal strength also varies depending on the concentration of tagged molecules. Some fluorescence systems, for example, are designed to detect the emission signal from a single molecule.
Fluorescence Filter Sets
Three types of optical filters are required for epi-fluorescence and other standard fluorescence microscopes to function properly: (1) an excitation filter, (2) an emission filter, and (3) a dichroic beamsplitter (Figures 1 & 4). Excitation and emission filters are bandpass filters that transmit a wavelength range that corresponds to the respective fluorophore absorption or emission spectrum, while blocking out extraneous light on either side. However, if image brightness is desired over contrast, emission filters can also be longpass edge filters that transmit longer wavelengths instead of blocking them. Most dichroic beamsplitters are longpass filters that are designed for use at a 45° angle of incidence (AOI). Dichroic filters direct the filtered excitation light to the sample, and the emission light from the sample through the emission filter to the detector (Figure 1).
Fluorescence filter sets can be either single or multi-band and can exist in multiple configurations, with the most common being fluorescence filter cubes and filter wheels. Single band filter sets can be used to view samples tagged with more than one fluorophore by taking a series of images with each respective filter cube, and then creating a final composite image. This can also be accomplished by using Pinkel or Sedat filter wheel configurations. Pinkel filter sets consist of a multi-band emission filter and a polychroic filter that are used along with multiple single-band excitation filters. Sedat filter sets consist of a polychroic filter that is used along with multiple single-band excitation and emission filters.
Full-multiband filter sets consisting of a multi-band excitation filter, a multi-band emission filter, and a polychroic filter are also available and will produce a single image containing all fluorescent tags.
Fluorescence Filter Set Requirements
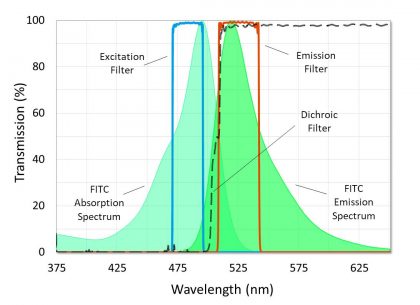
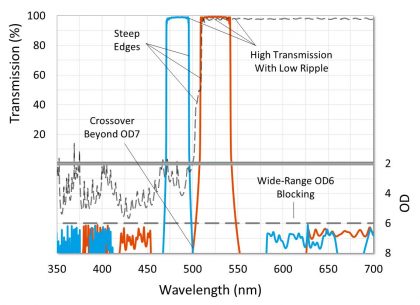
In order to construct a fluorescence filter set that produces a high quality image with a black background and high contrast, excitation and emission filters should achieve high transmission with minimal passband ripple over the regions corresponding to peak fluorophore excitation or emission, and their respective passbands should be separated so that overlap does not occur above a level of OD7 (-70dB or 0.00001% transmission) (Figure 5). However, a crossover of OD6 (-60dB or 0.0001% transmission) or OD5 (-50dB or 0.001% transmission) is acceptable when using a fluorophore with a very short Stokes shift, or in the case of some multiband filter sets.
Figure 5. Graph of a high-performance fluorescence filter set. Characteristics designed to give high contrast and a black background and are highlighted.
In order to achieve an OD7 crossover, fluorescence filters should be specified and designed as a set. Excitation and emission filters must also be designed with steep edges that transition from OD6 blocking to over 90% transmission in just a few nanometers in order to allow the passbands to capture the respective fluorophore peaks. Most fluorescence filter sets are designed so that the excitation and emission filter theory traces cross beyond OD7 to account for manufacturing tolerance. Because the measured spectra of coated filters can deviate from theory, it is also important to specify these filters in a way that ensures an appropriate crossover. Examples of this would be specifying a relatively tight center wavelength tolerance, or specifying absolute transmission or blocking ranges instead of average.
To achieve a black background, excitation and emission filters should be designed to block out any extraneous light. This is accomplished by specifying wide-range out-of-band blocking ranges at a level of OD6 or greater. Systems that utilize broadband light sources require blocking ranges from ~300 nm to ~1100 nm out-of-band, while switchable LED light sources require out-of-band blocking from ~300 nm to ~850 nm. Emission filters for laser systems require at least OD6 absolute blocking at the laser line, while out-of-band blocking ranges can be similar to emission filters for LED light sources. If ambient light is not a concern, then excitation filters for laser systems may not require wide-range blocking beyond the passband of the emission filter.
Dichroic filters are also required to have a relatively steep transition between reflection and transmission bands so the cut-on edge is placed between the passbands of the excitation and emission filters. In addition, all filters that transmit light to the detector or eyepiece should be specified with low transmitted wavefront error (TWE) in order to avoid image distortion.
Dichroic Flatness
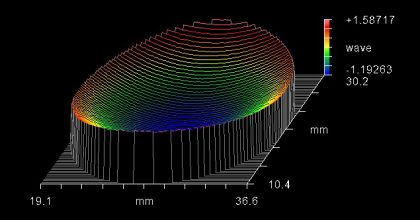
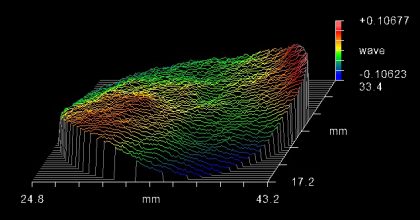
Surface flatness of the dichroic beamsplitter is a property that should be considered when selecting a fluorescence filter set for laser, imaging, total internal reflection fluorescence (TIRF), and super resolution systems. When a thin-film coating is deposited onto a substrate, the stress of the coating causes the substrate to bend, resulting in a bowl or dome shaped curvature (Figure 6). This coating-stress-induced curvature causes increased reflected wavefront error (RWE), which can result in focal shift and spot size distortion in these systems. Therefore, a low-stress coating (Figure 7) or backside compensation is necessary to avoid distortion.
Figure 6. Interferogram showing the curvature caused by coating stress that can result in image distortion.
However, challenging surface flatness specifications are generally not required for standard widefield epi-fluorescence microscopes that are equipped with a broadband lamp or LED light source. Most of these systems are configured so that the dichroic transmits the emission light to the detector, therefore TWE must be controlled but surface flatness is unlikely to affect the final image quality.
Zero Pixel Shift
Fluorescence filters should be specified to produce zero pixel shift when they are used in any configuration where the filters will be switched out in order to image multiple fluorophores within a single sample. These configurations include single-band filter sets, Pinkel sets, and Sedat sets. Emission filters and dichroic filters used in these configurations should be sourced from the same manufacturer and specified with a high degree of parallelism in order to minimize beam deviation caused by wedge angle. Because most filters are not perfectly parallel, and because coating stress will introduce a certain level of beam deviation, some degree of pixel shift will occur between images taken before and after optical filters are introduced into the system. However, when comparing images taken with zero pixel shift sets that were designed to work together, there should be a shift of less than approximately ± 1 pixel.
Minimizing Fluorophore Crosstalk
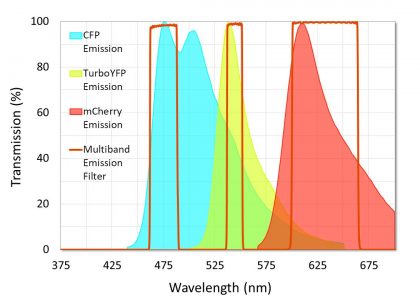
It is important to minimize crosstalk between fluorophores when using multi-band and Pinkel sets to image samples tagged with multiple fluorophores. Minimizing crosstalk is also an important consideration for fluorescent in situ hybridization (FISH), FRET, and flow cytometry applications. Crosstalk, or bleed-through, occurs when the excitation and emission signals of one fluorophore are transmitted through the filter channel reserved for a different fluorophore, resulting in a blending of colors in a single location that mimics the effect of co-localization. Although a certain amount of crosstalk is unavoidable, it is important to choose fluorophores and filter sets that minimize this effect. This can be accomplished by avoiding fluorophore combinations that have a large amount of overlap between their respective emission spectra. Overlap between absorption spectra should also be avoided in the cases where each fluorophore is excited by a different light LED or laser. Regardless of fluorophore selection, excitation and emission filters should be designed with relatively narrow passbands that capture the peak excitation or emission of the target fluorophore, while blocking as much of the overlap as possible (Figure 8).
Figure 8. Example of a multiband emission filter that was designed to minimize crosstalk between fluorophores. Note that crosstalk in the second channel is unavoidable because of the emission spectra overlap between CFP and TurboYFP.
Minimizing GDD
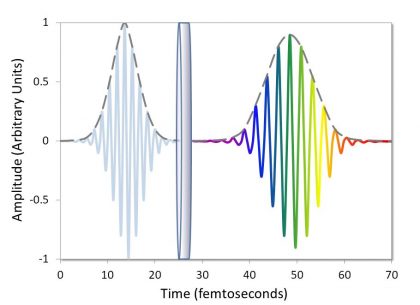
Non-linear optical (NLO) systems that use a pulsed femtosecond laser for excitation require optics that control phase shift and group delay dispersion (GDD). In these instruments, the pulsed beam is transmitted through or reflected off of several different optical components before ultimately reaching the sample. If these optical filters and mirrors are not specifically designed for use with femtosecond lasers, then the peak pulse intensity will decrease due to GDD every time the pulse train is reflected or transmitted (Figure 9). Because a reduction in peak pulse intensity will reduce the number of NLO signals, this compounded effect will ultimately result in poor instrument performance.
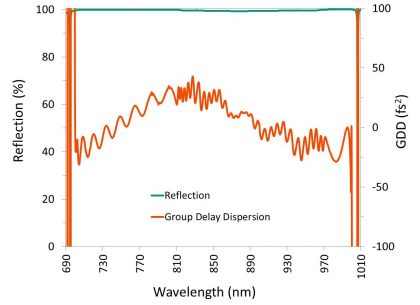
Dispersion-controlled dichroic filters and mirrors allow for simultaneous control of both the phase and amplitude of laser light (Figure 10), resulting in minimal dispersion and minimal loss of peak pulse intensity.
Alluxa fluorescence filters are all hard-coated using an innovative SIRRUS plasma deposition process. This process allows us to produce high-performance excitation, emission, and dichroic filters with steep edges, transmission that is typically greater than 95%, precision wavelength control, deep blocking, and low TWE, that are also specified to produce zero pixel shift.
Our dichroic beamsplitters for laser, imaging, TIRF, and super resolution applications are able to achieve flatness levels of λ/2 P-V per inch or better on 1.05 mm thick substrates, and levels of λ/10 P-V per inch or better on thicker substrates. We also offer high-reflectivity mirrors and dichroic filters that control GDD over a wide wavelength range.
Whether you are designing an OEM fluorescence system or are in the market for a filter cube, our knowledgeable engineers will work with you to design the fluorescence filters that best fit your specific requirements.
Why Alluxa?
Alluxa’s team of experts has delivered key innovations to the field of optical thin-films. In addition to designing and constructing all of our own custom optical thin-film coating equipment, we invented a novel plasma deposition coating process that both increases the performance of our optical filters and decreases the time it takes to produce them.
By combining these innovations with state-of-the-art automation, proprietary control algorithms, and precision monitoring during the coating process, we are able to deliver low-cost, high-performance, custom thin-film optical filters for any application.